News
Dec 06, 2025
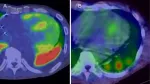
Carbon-11 Pittsburgh compound B (C-11 PiB) PET/CT demonstrates robust capability for detecting cardiac amyloidosis and accurately differentiating its subtypes,…
Nov 25, 2025
A Donegal man who was one of the first ever patients on an Amyloidosis treatment trial says the therapy has transformed his health.James Green, an accountant from…
Nov 17, 2025
Dr. Sami Khella, Professor of Neurology and co-founder of the Amyloidosis Program at the University of Pennsylvania discussed the latest in diagnosis, treatment,…
Oct 18, 2025
This webinar “Coffee Chat” style webinar is your chance to learn about the different programs and projects ARC has been working on in 2025! Hear directly from ARC…
Oct 17, 2025
Clinical Takeaway: Two studies show that cardiac amyloidosis is more common than we thought.ContextCardiac amyloidosis is traditionally considered to be relatively…
Oct 16, 2025
At Porterhouse, we are proud to support World Amyloidosis Day. Our article shines a light on this rare condition to help drive greater understanding, improve…
Oct 15, 2025
Clinical Psychologist Rosalind Kalb, PhD shared helpful advice, self-care tips, and real-life strategies to make daily life a little easier for both Care Partners…
Oct 14, 2025
Mohamad Mohty, Head of the Clinical Hematology and Cellular Therapy Department at Saint-Antoine Hospital, Sorbonne University, shared a post on X:“Join us for this…
Oct 07, 2025
AbstractTransthyretin (TTR) amyloidosis manifests in two distinct forms: hereditary (ATTRv) and wild-type transthyretin amyloidosis (ATTRwt). Despite being one of…
Oct 02, 2025
For many living with ATTR-CM amyloidosis, recent advances have brought hope. In just seven years we have gone from no approved treatments to having a number of…